S. Andonova, P. Kirilova, E. Kalevska, M. Petkova, Ts. Tsvetkov, M. Novakova, T. Georgiev
Neurosonology and Cerebral Hemodynamics 10, 2014:33–37
| TITLE | Decompressive Craniectomy in Ischemic Stroke – a Case Report |
| AUTHORS | S. Andonova, P. Kirilova, E. Kalevska, M. Petkova, Ts. Tsvetkov, M. Novakova, T. Georgiev |
| PUBL. DATE | May 2014 |
| SOURCE | Neurosonology and Cerebral Hemodynamics 10, 2014:33–37 |
| TYPE | Periodic scientific journal |
| ABSTRACT |
Objective: To discuss the therapeutic possibilities for treatment of malignant infarction after occlusion of M1 portion of the Middle Cerebral Artery (MCA). |
| KEY WORDS |
decompressive craniectomy, stroke |
Development of malignant cerebral infarction in distal portion of the internal carotid artery or proximal middle cerebral artery thrombosis is a therapeutic challenge as the massive cerebral ischemia limits the effectiveness of medication therapy. In cases of supratentorial infarctions resulting in death, the autopsy shows severe cerebral edema leading to herniation of cerebral structures through the tentorium or falx and brainstem structures through the foramen magnum. Transtentorial herniation has been cited as the probable cause of death in many cases of malignant stroke [4, 7, 9]. The clinical picture is characterized by changes in consciousness, progressive focal neurologic symptoms and brainstem dysfunction. Neuroimaging studies support the clinical picture. Extensive MCA infarction (more than 50% of its territory) with edema can be identified on CT scans in approximately 70% of the cases [19, 21]. Parenchymal hypodensity in greater than 50% of MCA territory is highly indicative for a progressive clinical course, leading to severe disability or death. It’s assumed that signs for extensive MCA infarction are: hyperdensity of the MСА, parenchymal hypodensity in greater than 1/3 of the MCA territory, hypodensity of basal ganglia. Clinical trials in patients with malignant MCA infarction show that decompressive craniectomy is life-saving and can significantly reduce mortality [1, 13]. Diagnosis is based on clinical and CT/MRI changes. The presented clinical case shows the therapeutic possibilities of the decompressive craniectomy and raises questions about this procedure’s timing and indications. It’s considered that the early treatment is linked with a better prognosis, although the optimal time window for performing surgery is still debated [5, 6, 8, 11].
Material and Methods
A 35-year-old male admitted for the first time in Second Neurology Clinic in the University Hospital “St. Marina” – Varna with sudden weakness of the left extremities based on high blood pressure was examined. There were many risk factors for cerebrovascular disease: arterial hypertension, dyslipidemia, obesity. A pointed laboratory examination of complete blood count, biochemistry and coagulation status was performed. Main head arteries were examined with Sonix SP (Canada) by color coded duplex scanning using 7.5 Hz transducer. The thickness of the carotid artery intima-media complex was measured by B-mode imaging in real-time using a standard program for automatic averaging of values. With pulse Doppler sonography speed parameters of blood flow were measured. Neuroimaging examination of the brain was conducted by 1.5 Tesla MRI (GE HTX Sigma – USA). An ischemic stroke in the territory of the MCA was diagnosed and confirmed by CT scan of the brain. All criteria of the National Health Insurance Fund for thrombolytic therapy were met and the patient underwent intravenous thrombolysis with rt-PA by protocol.
Results
In admission the patient was conscious, with an acute left-sided hemiplegia. The laboratory blood results were normal except for dyslipidemia. In absence of contraindications and after signing an informed consent from the patient's relatives, intravenous thrombolysis was performed. In the next 24 hours after the thrombolysis there was no change in the focal neurological deficit. Emergency MRI and magnetic resonance angiography (MRA) were performed because of worsening signs of increased intracranial pressure, showing persisting ischemia in the MCA territory, severe cerebral edema and signs of haemorrhagic infarction (fig. 1). On the third day after the symptoms onset and following a neurosurgical consultation, the patient underwent decompressive craniectomy. In the next few days the patient’s condition improved with full regression of edema but persisting severe left central hemiparesis to hand monoplegia (Rankin score 5). An improvement of focal neurological symptoms after three months to mild central left-sided hemiparesis was reported (Rankin score 3).
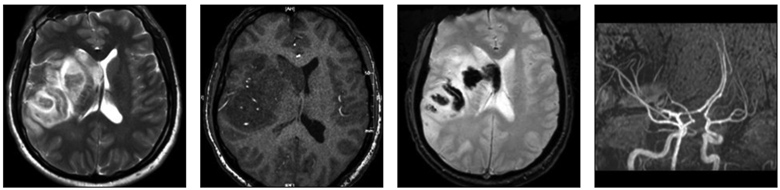
Fig. 1. MRI of the brain – ischemic stroke in MCA territory with hemorrhagic transformation and dislocation on the left.
Discussion
Malignant cerebral ischemia occurs in a significant number of patients with acute cerebrovascular incidents. The mortality rate in those patients is very high due to progressive, severe cerebral edema. In these patients decompressive craniectomy is indicated and is applied after an exact clinical and neuroimaging evaluation. The optimal treatment of brain edema includes osmotherapy with mannitol, blood pressure control, head elevation to 30°, maintenance of normothermia, normoglycemia and normovolemia. It’s considered that the early decompressive craniectomy leads to better results and is life-saving in some patients. The outcome of the decompressive craniectomy depends on procedure complications: insufficient decompression, infection, hemorrhage and development of contralateral cerebrospinal fluid collection [12, 14]. Advanced age, more severe motor deficit, longer duration of intensive therapy and prolonged mechanical ventilation are associated with a worse prognosis and quality of life of these patients [2, 3, 16, 18].
In conclusion the surgical decompression within the first 48 hours after the stroke onset reduces significantly the risk of death and severe disability in patients with malignant infarction due to occlusion of M1 portion of the MCA [72, 34, 30]. Questions about the indications and time window for this procedure remain open. Future studies are needed to focus on the long-term quality of life and neurological deficit in survivals.
REFERENCES
1. Akins PT, Guppy KH. Sinking skin flaps, paradoxical herniation, and external brain tamponade: a review of decompressive craniectomy management. Neurocrit Care 9, 2008: 269–276.
2. Arac A, Blanchard V, Lee M, Steinberg GK. Assessment of outcome following decompressive craniectomy for malignant middle cerebral artery infarction in patients older than 60 years of age. Neurosurg Focus 26, 2009:E3 1–6.
3. Arnaout OM, Aoun SG, Batjer HH, Bendok BR. Decompressive hemicraniectomy after malignant middle cerebral artery infarction: rationale and controversies. Neurosurg Focus 30, 2011:E18 1–5.
4. Bounds JV, Wiebers DO, Whisnant JP; Mechanism and timing of deaths from cerebral infarction. Stroke 12, 1981: 474–477.
5. Carter BS, Ogilvy CS, Candia GJ. One-year outcome after decompressive surgery for nondominant hemispheric infarction. Neurosurg 40, 1997:1168–1176.
6. Chang V, Hartzfeld P, Langlois M, Mahmood A, Seyfried D. Outcomes of cranial repair after craniectomy. J Neurosurg 112, 2010:1120–1124.
7. Danish SF, Barone D, Lega BC, Stein SC. Quality of life after hemicraniectomy for traumatic brain injury in adults. A review of the literature. Neurosurg Focus 26, 2009: E2 1–5.
8. Doerfler A, Forsting M, Reith W, Staff C, Heiland S, SchaЁbitz WR, von Kummer R, Hacke W, Sartor K. Decompressive craniectomy in a rat model of “malignant” cerebral hemispherical stroke: experimental support for an aggressive therapeutic approach. J Neurosurg 85, 1996:853–859.
9. Engelhorn T, Doerfler A, Kastrup A. Decompressive craniectomy, reperfusion, or a combination for early treatment of acute “malignant” cerebral hemispheric stroke in rats? Potential mechanisms studied by MRI. Stroke 30, 1999: 1456–1463.
10. Foerch C, Lang JM., Krause J, Raabe A, Sitzer M, Seifert V, Steinmetz H, Kessler KR. Functional impairment, disability, and quality of life outcome after decompressive hemicraniectomy in malignant middle cerebral artery infarction. J Neurosurg 101, 2004:248–254.
11. Forsting M, Reith W, Schaebitz WR. Decompressive craniectomy for cerebral infarction: an experimental study in rats. Stroke 26, 1995:259–264.
12. Frank JI, Krieger D, Chyatte D. Hemicraniectomy and durotomy upon deterioration from massive hemispheric infarction: a proposed multicenter, prospective, randomized study. Stroke 30, 1999:243.
13. Gupta R, Connolly ES, Mayer S, Elkind MS. Hemicraniectomy for massive middle cerebral artery territory infarction: a systematic review. Stroke 35, 2004:539–543.
14. Hacke W, Schwab S, Horn M, Spranger M, De Georgia M, von Kummer R. Malignant middle cerebral artery territory infarction: clinical course and prognostic signs. Arch Neurol 53, 1996:309–315.
15. Heros RC. Surgical treatment of cerebellar infarction. Stroke 23, 1992:937–938.
16. Hofmeijer J, Kappelle LJ, Algra A, Amelink GJ, van Gijn J, van der Worp HB. Surgical decompression for space-occupying cerebral infarction (the Hemicraniectomy After Middle Cerebral Artery infarction with Life-threatening Edema Trial [HAMLET]): a multicentre, open, randomised trial. Lancet Neurol 8, 2009:326–333.
17. Jourdan C, Convert J, Mottolese C. Evaluation of the clinical benefit of decompression hemicraniectomy in intracranial hypertension not controlled by medical treatment. Neurochirurgie 39, 1993:304–310.
18. Jattler E, Bцsel J, Amiri H, Schiller P, Limprecht R, Hacke W, Unterberg A. DESTINY II: DEcompressive Surgery for the Treatment of malignant INfarction of the middle cerebral arterY II. Int J Stroke 6, 2011:79–86.
19. Kasner SE, Demchuk AM, Berrouschot J, Schmutzhard E, Harms L, Verro P, Chalela JA, Abbur R, McGrade H, Christou I, Krieger DW. Predictors of fatal brain edema in massive hemispheric ischemic stroke. Stroke 32, 2001:2117–2123.
20. Krieger DW, Demchuk AM, Kasner SE, Jauss M, Hantson L. Early clinical and radiological predictors of fatal brain swelling in ischemic stroke. Stroke 30, 1999:287–292.


